Heavy metal contamination resulting from industrial activities such as mining, smelting, and electroplating has become a global ecological and environmental challenge. Birnessite, a manganese oxide mineral widely distributed in soils and sediments, has an exceptional capacity to adsorb heavy metals and is often regarded as a “scavenger” of heavy metals. It is therefore a key mineral controlling the migration and transformation of heavy metals in geological media. However, birnessite is unstable and can readily undergo phase transformation in natural environments, potentially releasing previously adsorbed heavy metals and creating environmental risks. Previous studies have shown that microorganisms can induce birnessite transformation under natural conditions, but the specific mechanisms by which this process affects heavy metal migration and transformation remain poorly understood.
Focusing on birnessite as the target mineral and Pb(II) as the representative contaminant, this study employed the naturally widespread bacterium Shewanella putrefaciens CN32 to induce birnessite transformation under both aerobic and anaerobic conditions. Batch adsorption experiments of Pb(II) were then conducted on the transformation products. By integrating X-ray diffraction, Raman spectroscopy, X-ray photoelectron spectroscopy, extended X-ray absorption fine structure analysis, and surface complexation modeling, the researchers systematically investigated how microbially induced birnessite transformation affects both the adsorption capacity and adsorption mechanisms of Pb(II). The study led to the following major findings:
Under aerobic conditions, CN32 partially reduced Mn(IV) to Mn(II/III). Although the overall mineral phase of birnessite remained unchanged, the number of Mn vacancies and highly reactive surface sites decreased, causing the Pb(II) adsorption capacity to drop from 598 mg/g to 323 mg/g. However, this localized reduction also generated Mn(III)-coordinated oxygen with stronger electron-donating ability, which increased the covalency of the Pb–O bond and, in turn, enhanced the environmental stability of Pb(II).
Under anaerobic conditions, birnessite underwent a dissolution–reprecipitation process and was transformed into secondary minerals including manganite (MnOOH), hausmannite (Mn3O4), and rhodochrosite (MnCO3). Because these secondary minerals inherently possess lower adsorption capacities, the Pb(II) adsorption capacity of the transformed products decreased sharply from 598 mg/g to 129 mg/g, while the environmental stability of Pb(II) also declined.
The results demonstrate that microbial-induced birnessite transformation not only changes the adsorption capacity of birnessite for Pb(II), but also alters the environmental stability of Pb(II) by modifying its surface coordination mode. The study provides deeper insight into the migration and transformation of heavy metals in geological media and offers a theoretical basis for the development of scientifically sound strategies for heavy metal risk prevention and control at mining areas and contaminated sites.
The findings were recently published in Geochimica et Cosmochimica Acta under the title “Microbial-induced birnessite transformation under different redox conditions and its impact on lead adsorption.” Doctoral student Yuanyuan Geng from the School is the first author, and Associate Professor Zhengtao Shen is the corresponding author. The co-authors are Professor Bin Shi, Professor Chaosheng Tang, Professor Baojun Wang, Assistant Professor Huan Liu, doctoral student Xiaoliang Fan, doctoral student Guanyu Zhang, doctoral student Lingyi Tang from the University of Alberta, and Professor Daniel Alessi from The University of Texas at Austin.
This research was supported by the the National Key Research and Development Program of China (2023YFC3707900), the Fundamental Research Funds for the Central Universities (2024300399), National Natural Science Foundation of China (42277123, 42477187), Natural Science Foundation of Jiangsu Province (BK20220787).
Full paper:https://doi.org/10.1016/j.gca.2026.02.005
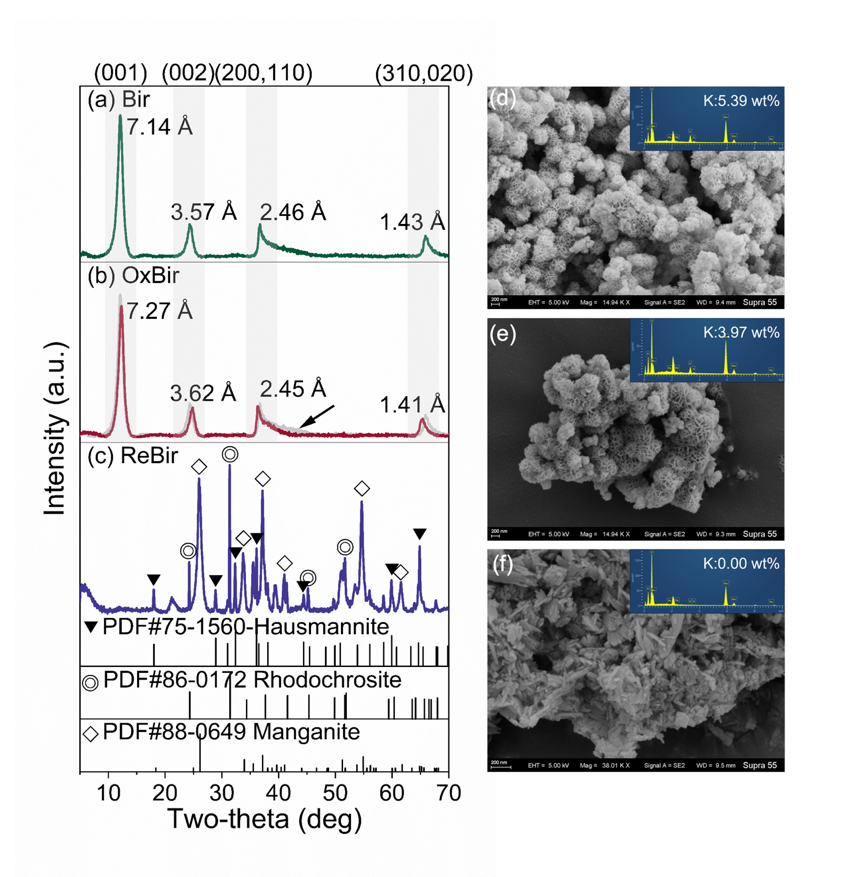
Fig. 1. XRD and SEM characterization of birnessite before and after microbial-induced transformation.
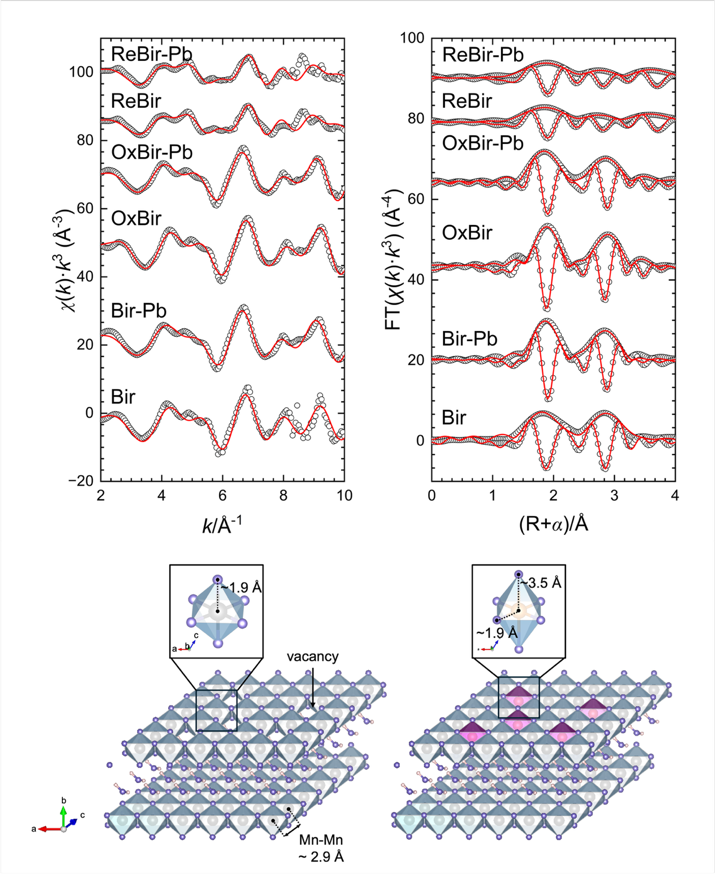
Fig. 2. Mn K-edge EXAFS spectra of the samples before and after Pb(II) adsorption.
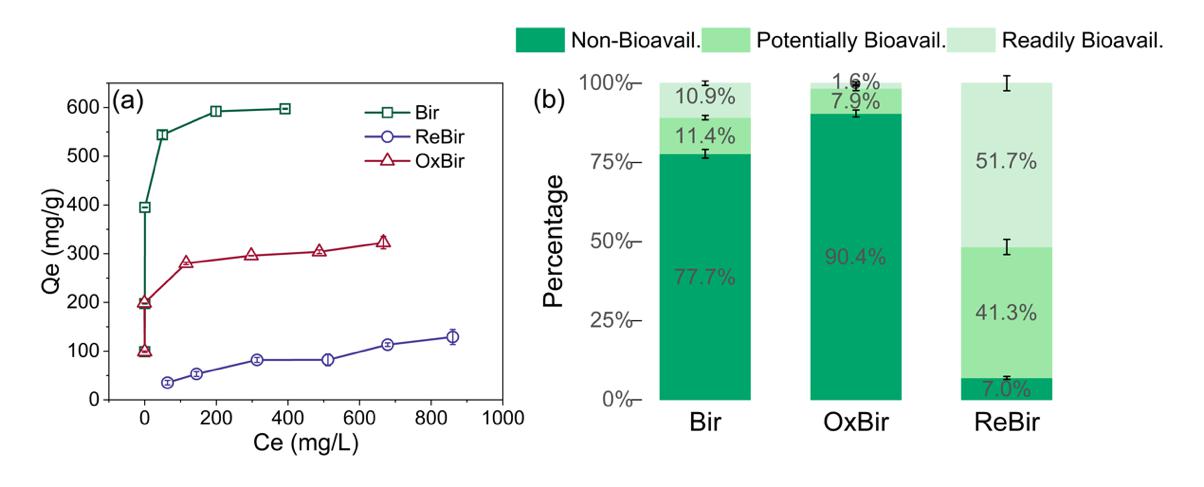
Fig. 3. Pb(II) adsorption isotherms on birnessite and Pb(II) speciation distribution by sequential extraction before and after microbially induced phase transformation.
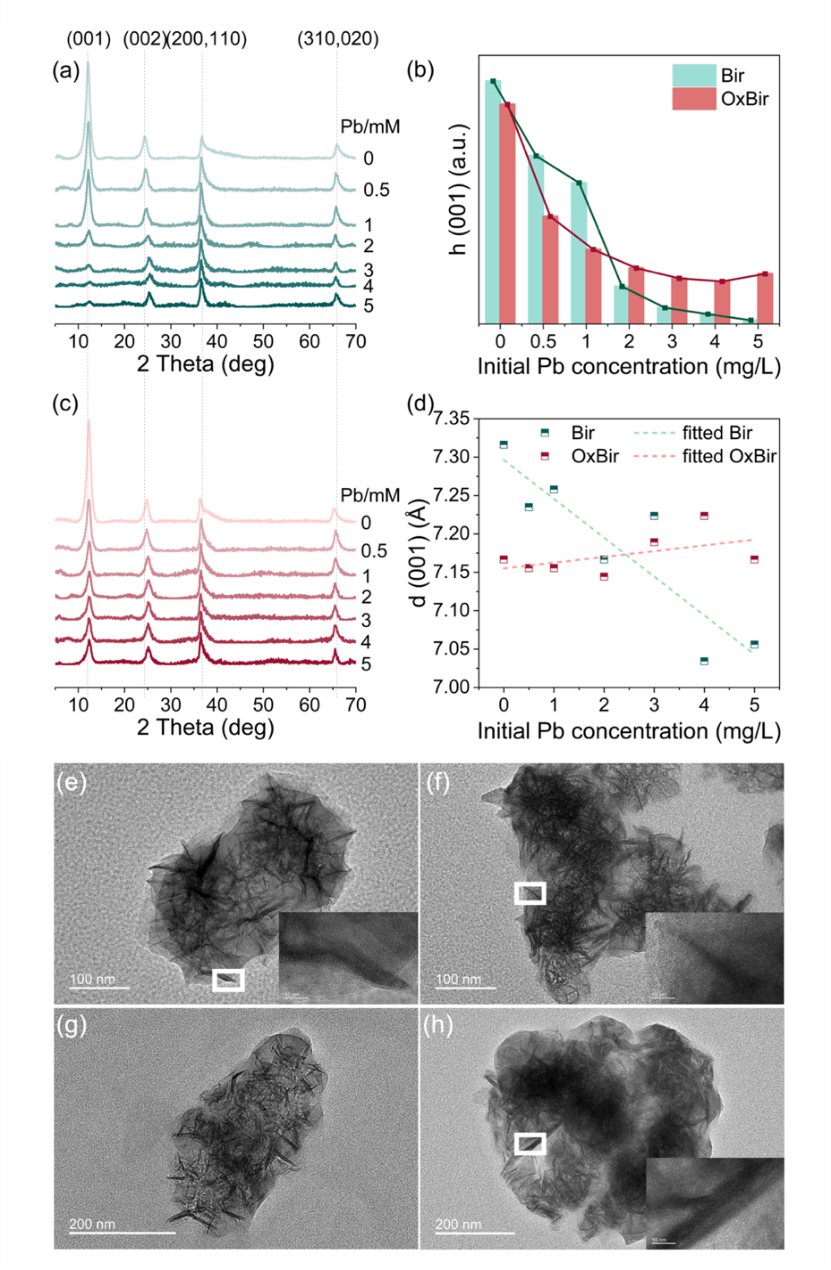
Fig. 4. XRD and TEM characterization results of birnessite before and after Pb(II) adsorption.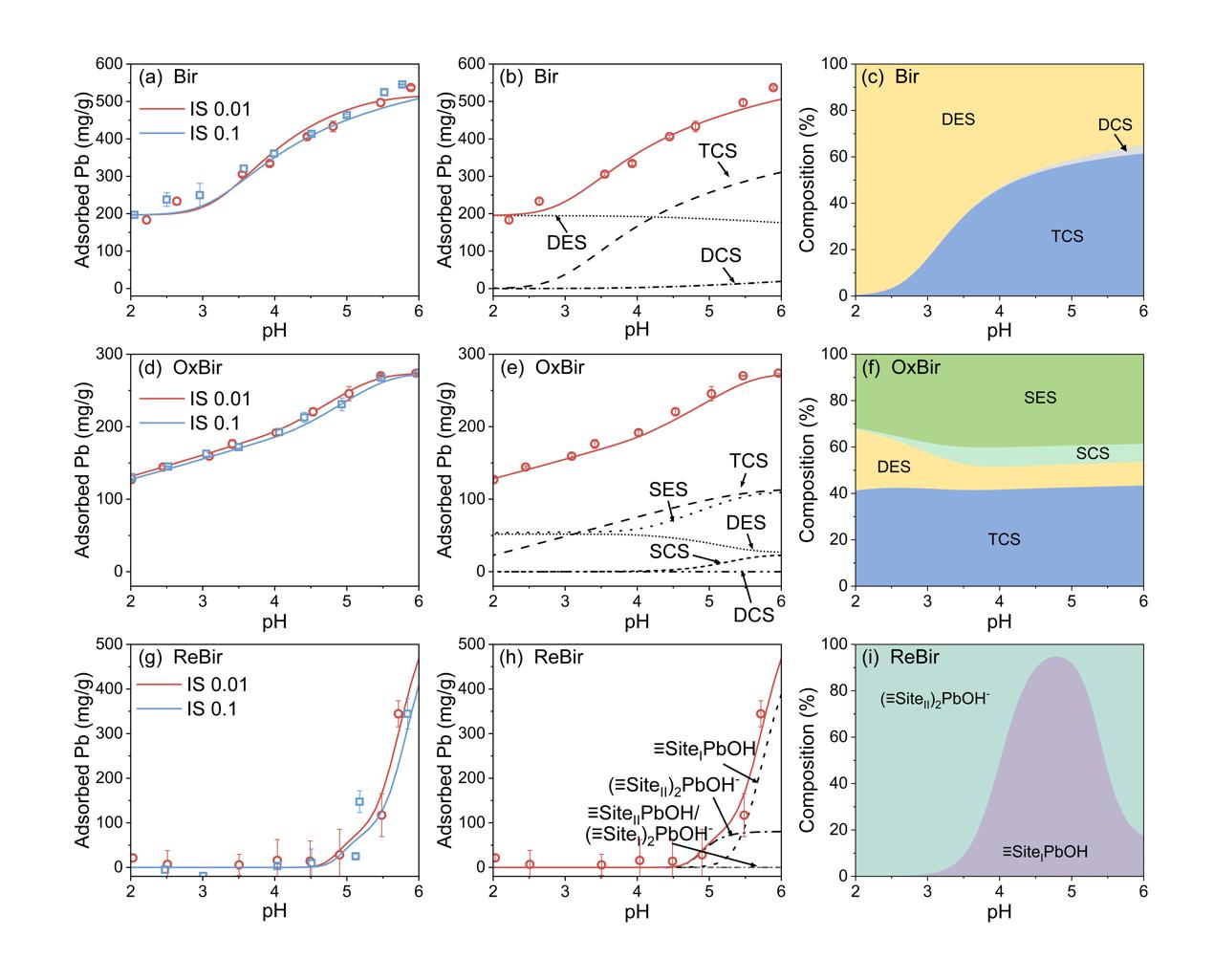
Fig. 5. Pb(II) adsorption edges on birnessite and Pb(II) speciation distribution predicted by the surface complexation model.

