
Lithium isotopes exhibit significant fractionation during low-temperature geological processes and are widely used as tracers for crustal recycling and subduction processes. Traditional views hold that lithium isotope fractionation is limited in high-temperature magmatic systems. However, recent observations of large δ7Li variations, ranging from −10‰ to +20‰ in lithium-cesium-tantalum (LCT) pegmatites, highlighting the need for a quantitative understanding of the controlling mechanisms. A key limitation lies in the absence of reliable fractionation factors for silicate melts, likely due to the complexities in melt structures, composition, and dynamics.
In this study, we use ab initio molecular dynamics (AIMD) simulations and density functional theory (DFT) calculations to determine the coordination geometries and lithium isotope fractionation factors among minerals, felsic melts and hydrothermal fluids (Figure 1). This study presents the first calculations of lithium isotope fractionation in felsic melts and applies the second-order vibrational perturbation theory (VPT2) method to both melt and fluid systems for the correction of anharmonic effects. By combining first-principles molecular dynamics, density functional theory, and numerical simulations, a multi-scale quantitative framework of lithium isotope fractionation among the phases of mineral-melt-fluid was constructed.
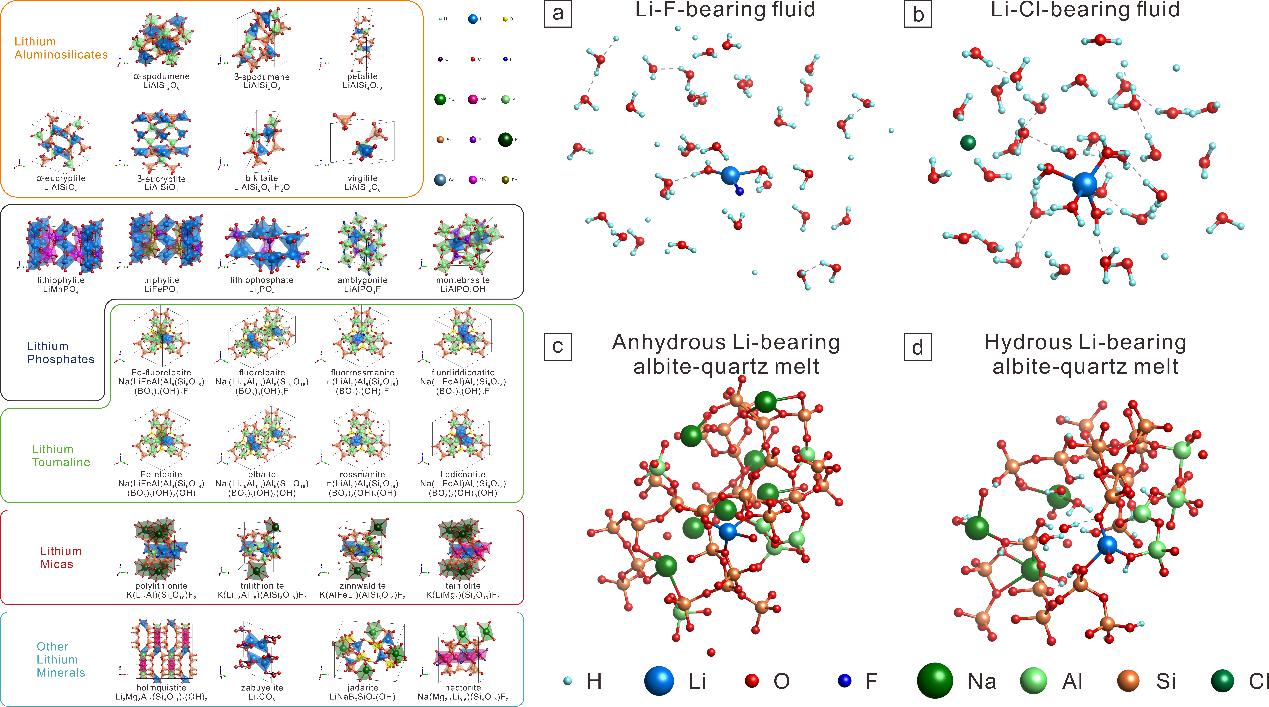
Figure 1 Representative configurations of Li-bearing minerals, hydrothermal fluids, and felsic melts
The numerical modelling outlined in this study provides a good description of the lithium isotope variations in fractional crystallization, fluid exsolution, melt-melt separation, as well as diffusion induced kinetic effects. The equilibrium fractionation during fractional crystallization is limited (−3.5 to +1.5‰) and therefore insufficient to account for the large isotopic variability observed in natural pegmatites (Figure 2). Melt-melt immiscibility generates moderate fractionation (~6‰), although the isotopic contrast between the immiscible melts diminishes during subsequent differentiation (Figure 3). In contrast, fluid exsolution produces the largest isotopic shifts, with melt-fluid fractionation exceeding 10‰ (Figure 4). Diffusion introduces an additional 3–8‰ kinetic fractionation within boundary layers and along melt–mineral interfaces (Figure 5).
In summary, the lithium isotope variations exceeding 10‰ observed in typical pegmatite deposits such as Jiajika (Eastern Tibet, China), Tin Mountain (USA), and Qinghe (Xinjiang, China) may be primarily controlled by fluid exsolution and diffusion-related kinetic effects. These findings enhance our understanding of lithium isotope behavior in pegmatitic systems and establish lithium isotopes as robust tracers for reconstructing magmatic-hydrothermal evolution and light new insights into petrogenesis and mineralization of pegmatites using lithium isotopes.
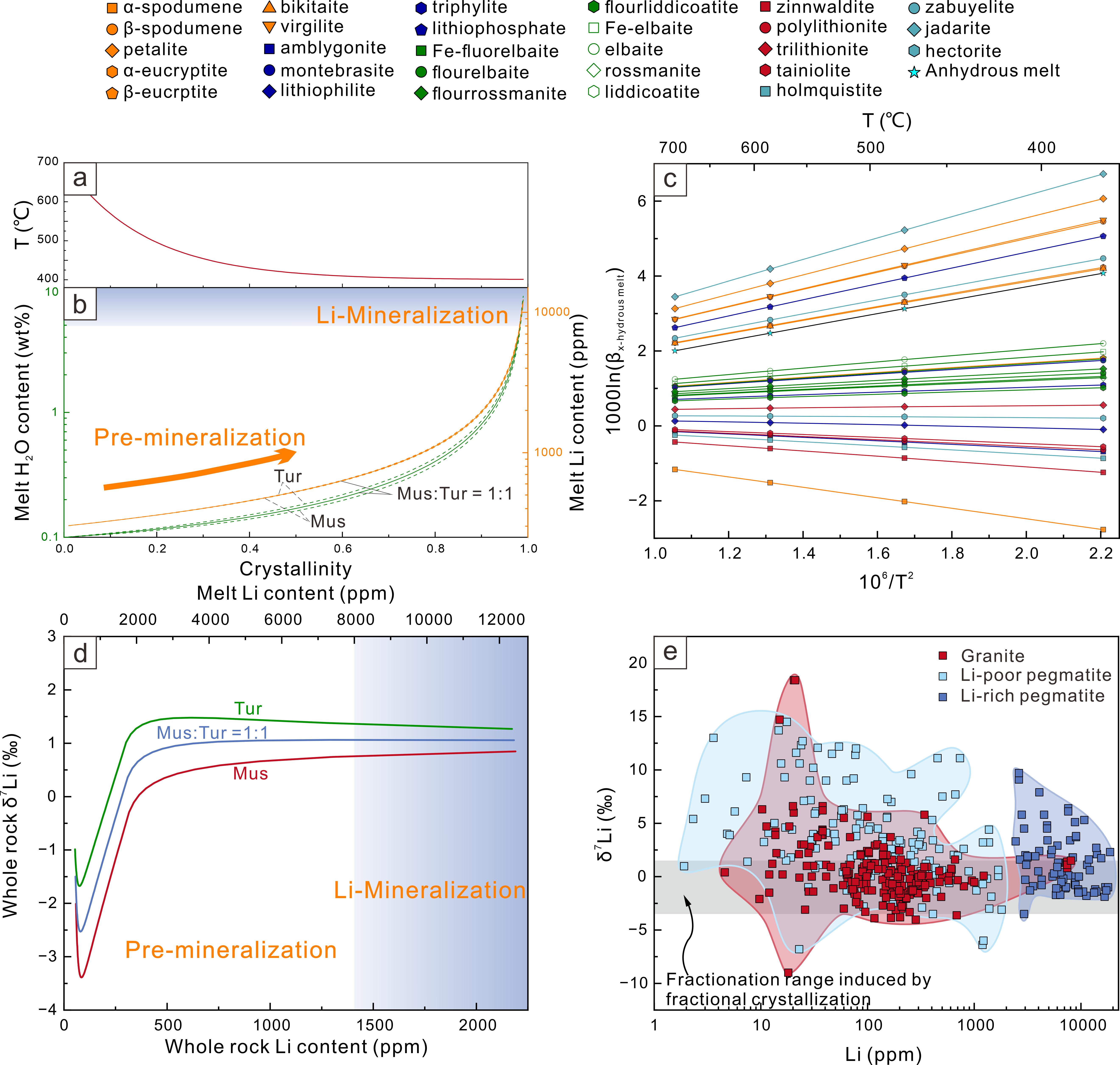
Figure 2 Li isotope fractionation during fractional crystallization.
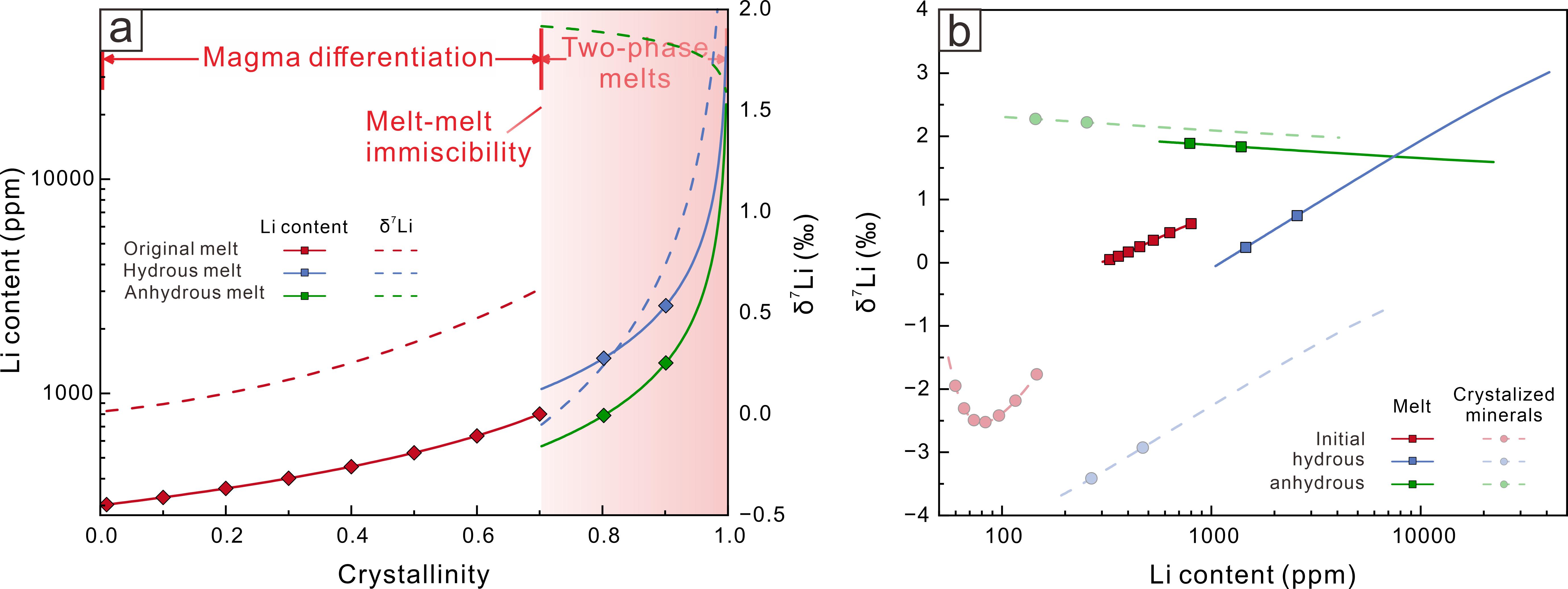
Figure 3 Li isotope fractionation during melt-melt immiscibility.
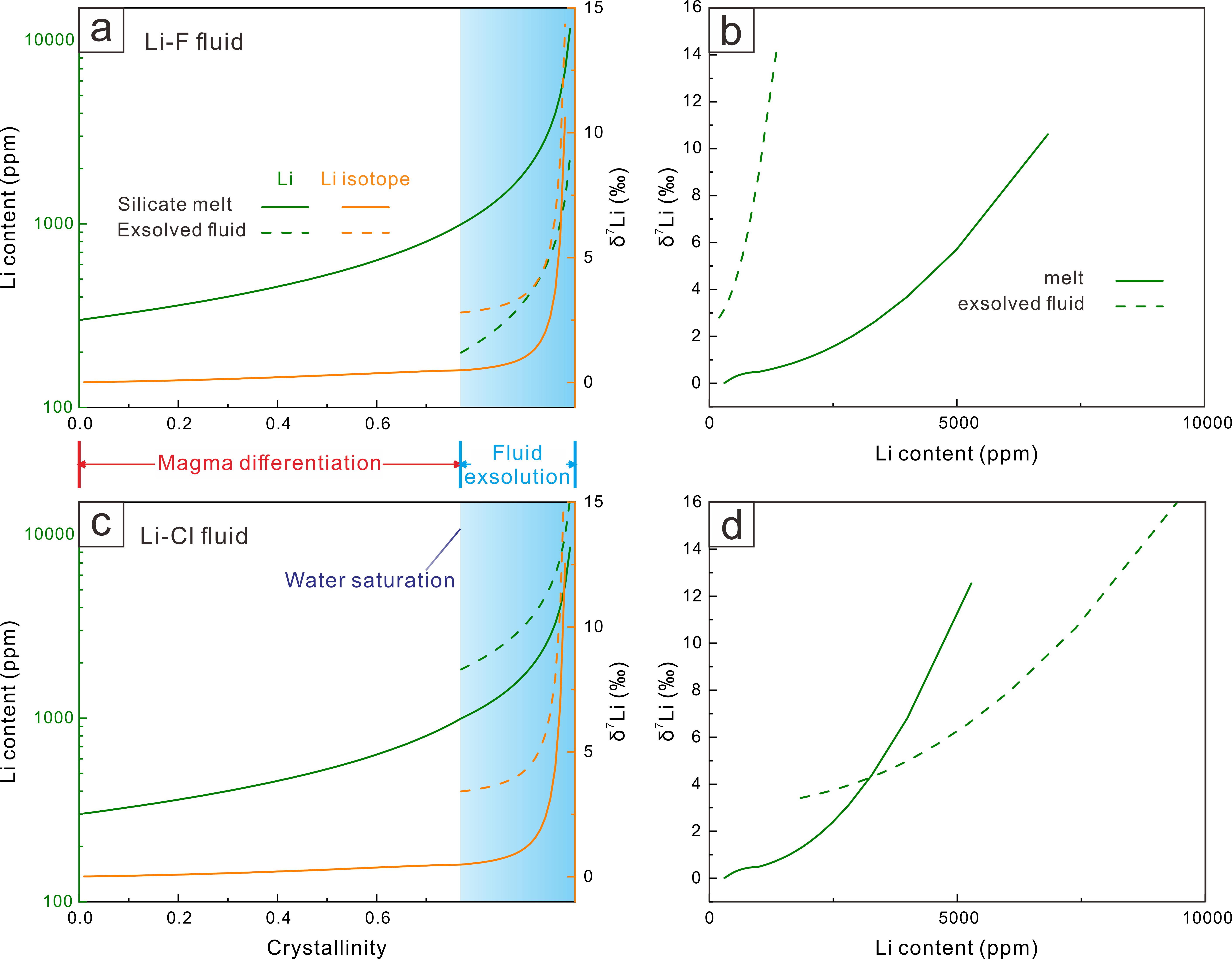
Figure 4 Li isotope fractionation during fluid exsolution.
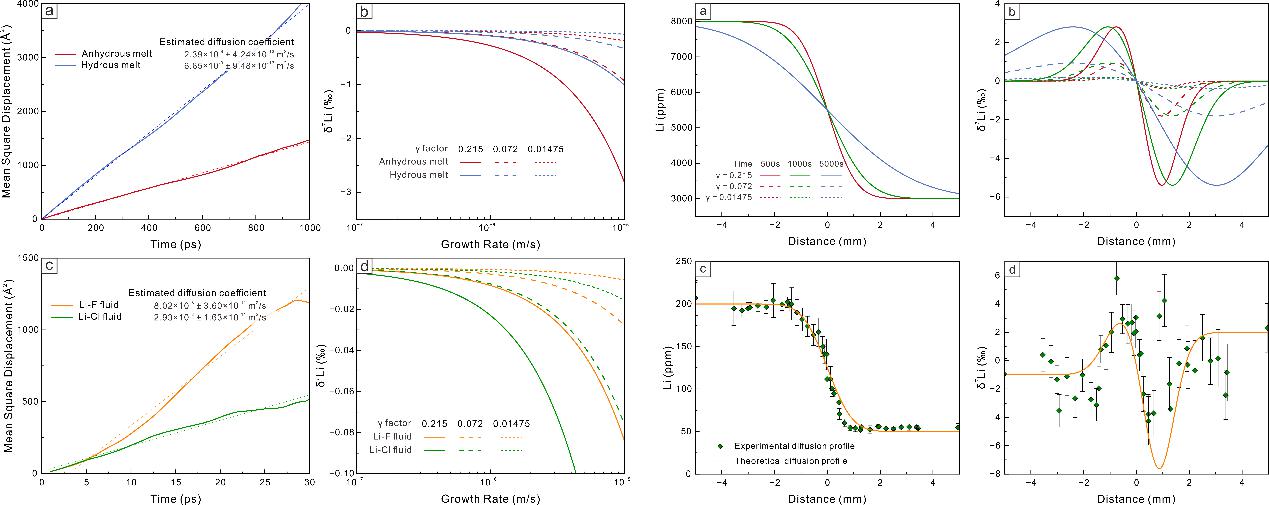
Figure 5 Numerical modeling of kinetic Li isotope fractionation in rapid crystal growth from silicate melts and hydrothermal fluids and in diffusion-controlled fractionation.
The research was published online on February 26, 2026, in Geochimica et Cosmochimica Acta (GCA). He-Feng Lin, a doctoral student from the School of Earth Sciences and Engineering, Nanjing University, is the first author of the paper. Professors Wen-Bin Zhu and Hai-Zhen Wei from the School of Earth Sciences and Engineering, Nanjing University, are the co-corresponding authors. Co-authors include Academician Zhi-Qin Xu and Professor Xi-Sheng Xu (School of Earth Sciences and Engineering, Nanjing University), Professor Martin R. Palmer (University of Southampton, UK), Professor Jing Ma (School of Chemistry, Nanjing University), Associate Professor Shuang Chen (Kuang Yaming Honors School, Nanjing University), Associate Professor Yin-Chuan Li (Henan Polytechnic University), and graduate students Da-Sheng Zuo and Ke Yang.
This study was funded by National Natural Science Foundation of China (92162211, 91955203, 41973005), Nanjing University Excellence Initiative ‘Scientific drilling for the pegmatitic lithium deposit in western Sichuan’, and an Open Research Fund (Grant No. 2025-Z05) of State Key Laboratory of Critical Earth Material Cycling and Mineral Deposits, Nanjing University.
Full article is available at: https://doi.org/10.1016/j.gca.2026.02.035

