Montmorillonite, a representative 2:1 type clay mineral, is among the most abundant minerals at the Earth's surface. Owing to their high specific surface area, large interlayer cation exchange capacity, and strong surface reactivity, they play a crucial role in governing the geochemical cycling of metallic elements and the environmental fate of heavy metals. Accurately predicting the competitive adsorption of multiple metals on clay mineral surfaces is therefore a key scientific issue for understanding the geochemical behavior of metal elements and for developing remediation materials for heavy-metal pollution. Conventional surface complexation models (SCMs) rely on empirical assumptions and the fitting of experimental data, without a rigorous theoretical framework to describe interfacial reaction mechanisms, resulting in limited transferability and extensibility. While capable of describing single-metal adsorption behavior, these models remain insufficient for accurately predicting competitive adsorption among multiple metals.
Researchers from the Computational Mineralogy Group at School of Earth Sciences and Engineering, Nanjing University, have developed a multiscale theoretical model to simulate competitive metal adsorption on clay mineral surfaces (Fig. 1).Grounded in First-Principles Molecular Dynamics (FPMD) calculations, the model integrates crystal structure and interfacial reactivity within a bottom-up framework, enabling precise determination of the geometric parameters and acidity of surface sites, as well as elucidation of metal uptake mechanisms, including isomorphic substitution, interlayer cation exchange, and competitive adsorption on edge surfaces (Fig. 2).The model successfully reproduces experimental data for multi-metal competitive adsorption systems (Fig. 3), demonstrating significantly enhanced predictive accuracy compared to the widely used two-site non-electrostatic surface complexation/cation exchange model (2SPNE SC/CE) developed at the Paul Scherrer Institute in Switzerland (Fig. 3A).Furthermore, the model captures the transitions among three adsorption mechanisms—cation exchange, independent adsorption, and competitive adsorption—under varying pH conditions (Fig. 3B), providing a quantitative description of metal partitioning on clay mineral surfaces.The model establishes a new theoretical framework for multiscale studies of elemental geochemical cycling and environmental behavior, offering significant potential for applications in metal mineralization, soil remediation, and nuclear waste repository safety assessment.
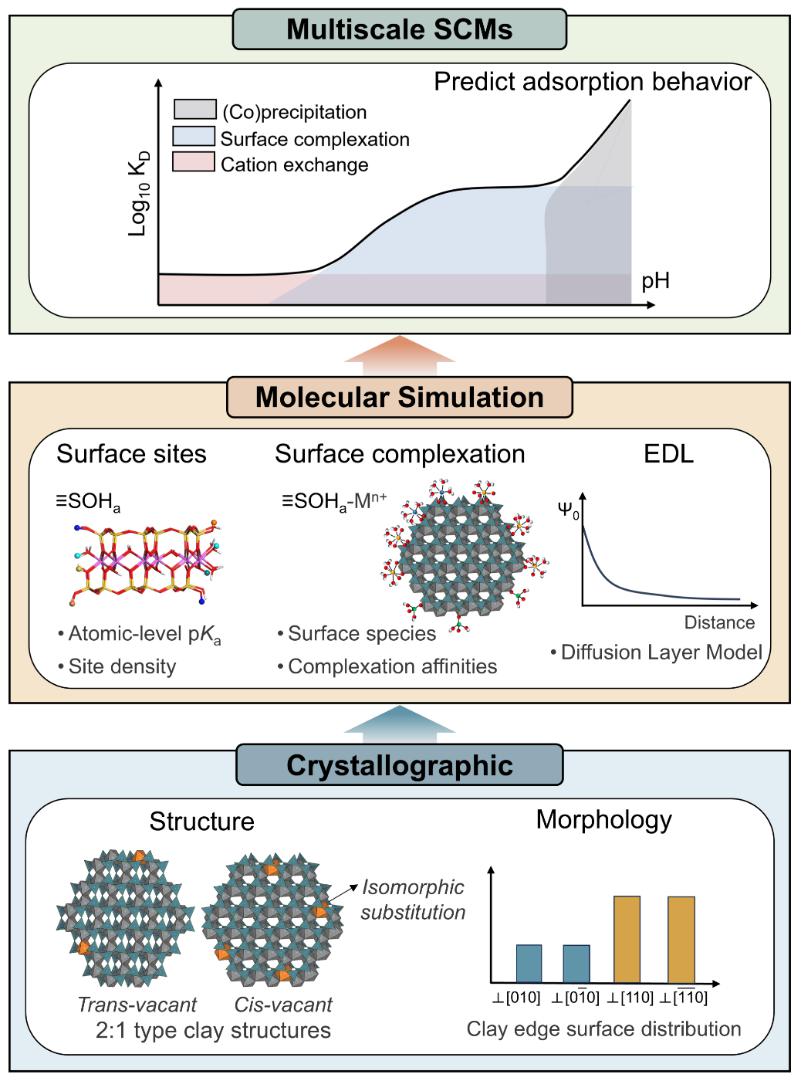
Figure 1. Development of multiscale surface complexation model for clay mineral surfaces.
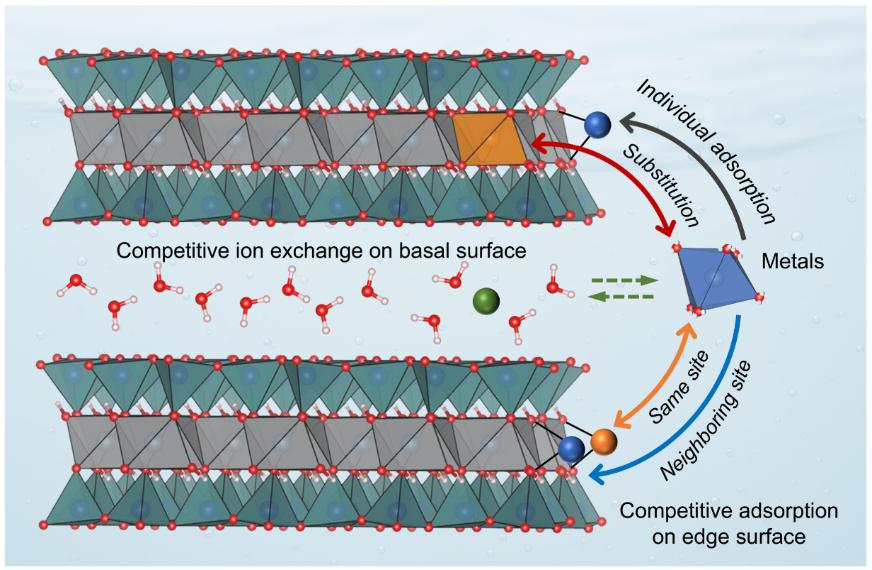
Figure 2. Schematic illustration of metal uptake mechanisms on montmorillonite.
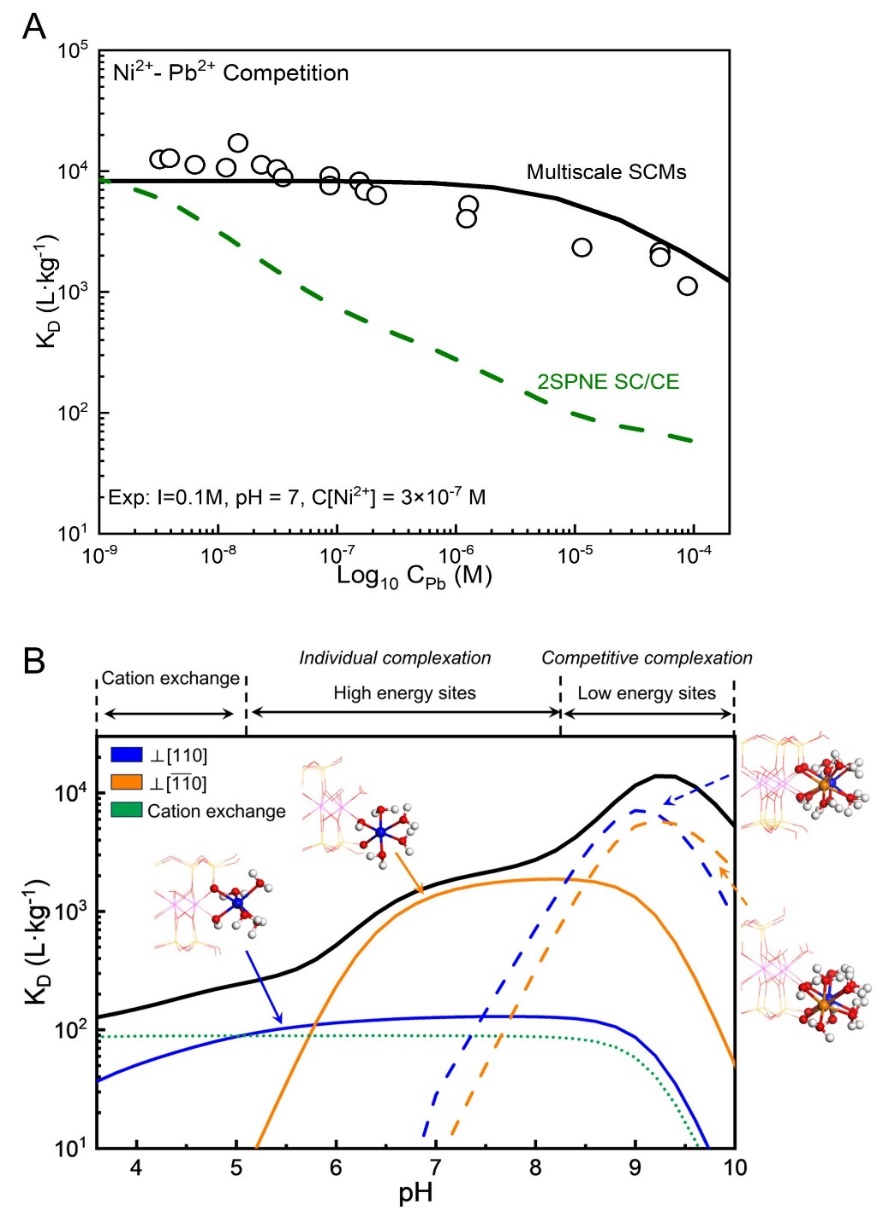
Figure 3. (A) Ni2+ adsorption curve on montmorillonite versus Pb2+ concentration under competitive adsorption; (B) Decomposition of Ni2+ adsorption curve on montmorillonite as a function of pH.
This work was published on February 24, 2026, in Proceedings of the National Academy of Sciences of the United States of America (PNAS) under the title "An Accurate and Efficient Framework for Modelling Multi-Metal Competitive Adsorption on Clay Minerals". The study was led by Pengyuan Gao, Ph.D. (first author), Prof. Xiandong Liu (corresponding author), and Prof. Xiancai Lu (co-author) are affiliated with the School of Earth Sciences and Engineering, the State Key Laboratory of Critical Earth Material Cycling and Mineral Deposits, and the Frontiers Science Center for Critical Earth Material Cycling at Nanjing University, with Prof. Christophe Tournassat (co-author) from ISTO, University of Orléans, France. This research was jointly supported by the National Natural Science Foundation of China, Fundamental and Interdisciplinary Disciplines Breakthrough Plan of the Ministry of Education of China, Postdoctoral Fellowship Program of CPSF, and China Postdoctoral Science Foundation.
Original article: https://www.pnas.org/doi/10.1073/pnas.2527567123

