Carbonatite magmas are a key carrier of the deep carbon cycle. Their chemical composition differs fundamentally from that of silicate magmas, which account for the vast majority of magmatic activity on Earth. This unusual composition has long attracted scientific interest and is also responsible for the remarkable enrichment of critical metals such as niobium (Nb), an element of growing importance due to its applications in high-technology and strategic industries.
Despite decades of research, the origin of extraordinary Nb enrichment in carbonatite systems remains debated. A central question concerns whether Nb mineralization is primarily controlled by magmatic differentiation or by hydrothermal processes. In most carbonatite-related deposits, niobium occurs mainly in pyrochlore ((Na,Ca)2Nb2O6(F,OH)), the principal Nb-bearing mineral and the dominant economic ore mineral in many carbonatite deposits.
In this study, controlled high-temperature and high-pressure experiments were conducted to systematically constrain the solubility and crystallization behavior of Nb in synthetic carbonatite melts (Fig. 1). The results provide the first experimental evidence for a magmatic differentiation–driven mechanism responsible for massive pyrochlore crystallization in carbonatite systems, and lead to the proposal of an integrated magmatic–hydrothermal model for Nb mineralization in carbonatites.
Experimental results show that Nb solubility in carbonatite melts decreases sharply with decreasing temperature and increasing Na content. During magmatic differentiation, the solubility of Nb (expressed as Nb2O5) can drop dramatically from several wt.% to below 0.03 wt.% (Fig. 2). Quantitative modeling indicates that this drastic decrease can trigger large-scale pyrochlore crystallization within a narrow temperature interval of approximately 700–750 °C (Fig. 3). This process represents a critical mechanism for the formation of Nb mineralization during the differentiation and crystallization of carbonatite magmas (Fig. 4).
These findings significantly advance our understanding of the petrology and geochemistry of carbonatite systems and provide a new genetic framework for the exploration of critical metal deposits. The results offer both fundamental scientific insights and practical implications for mineral exploration.
The study was recently published in Geochimica et Cosmochimica Acta, a Nature Index journal. The first author is Yating Xu, a PhD candidate at the School of Earth Sciences and Engineering, Nanjing University. Prof. Rucheng Wang is the corresponding author. Co-authors include Dr. Xudong Che (Nanjing University), Prof. Fabrice Gaillard, Dr. Marion Louvel, and Dr. Giada Iacono-Marziano (CNRS–ISTO), as well as Dr. Johann Tuduri (BRGM). This research was supported by the Key Program of the National Natural Science Foundation of China (Grant No. 42230809) and the China Scholarship Council Innovation Program (Grant No. CXXM2110080163).
Article information:
Xu, Y. T., Wang, R. C., Che, X. D., Gaillard, F., Louvel, M., Iacono-Marziano, G., & Tuduri, J. (2025). A dramatic drop in Nb solubility during fractional crystallization of carbonatites triggers Nb ore formation. Geochimica et Cosmochimica Acta.
https://doi.org/10.1016/j.gca.2025.10.021.
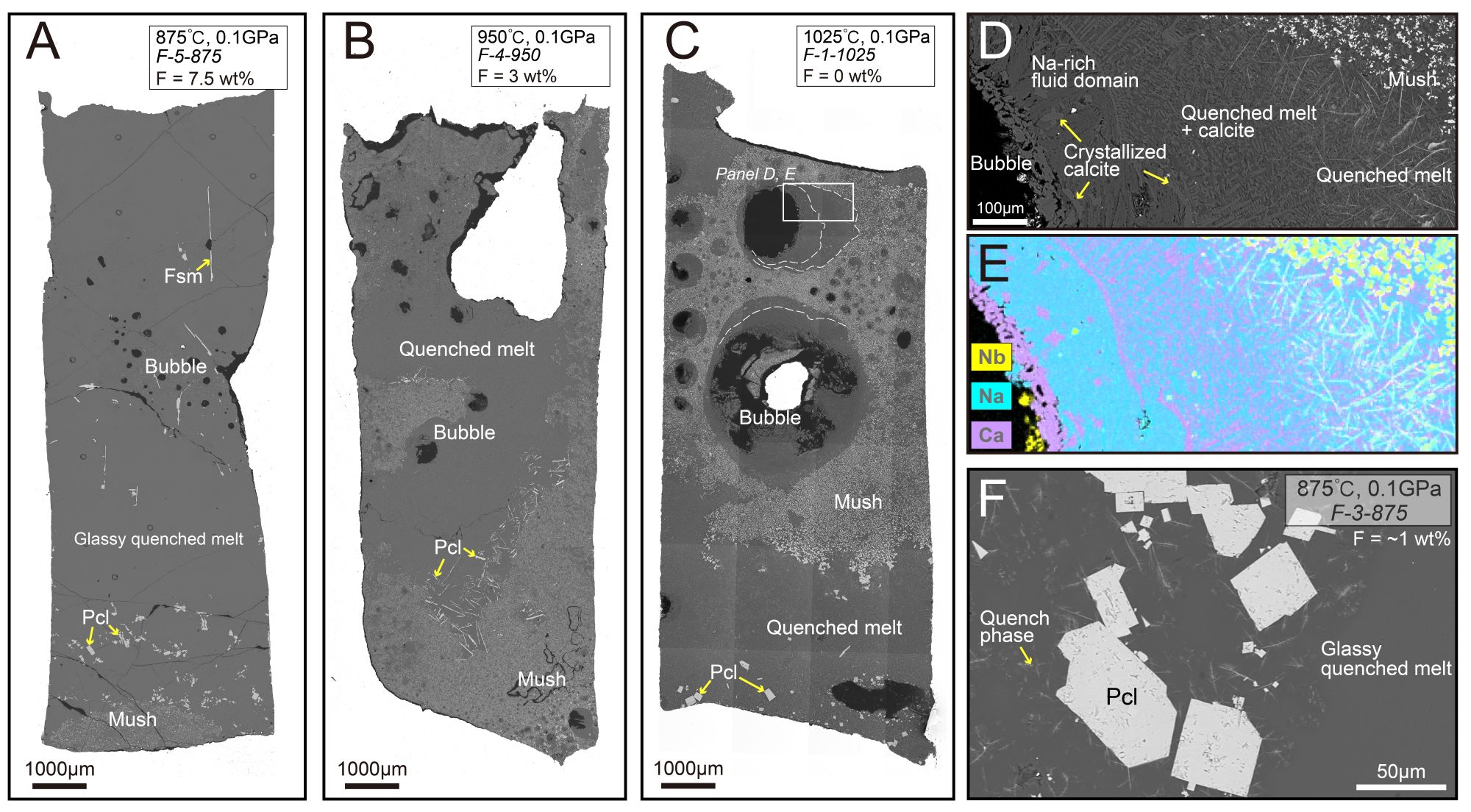
Figure 1. Characterization of experimental products. (A–C) Back-scattered electron (BSE) images of entire sample capsules, showing the morphology of experimental products formed under different temperature and fluorine conditions. (D) Magnified image of the experimental product at 1025 °C, showing the quenched melt and Na-rich fluid regions surrounding vesicles. (E) Energy-dispersive spectroscopy (EDS) elemental maps of the area shown in (D), illustrating the compositional distribution. (F) BSE image showing crystallized pyrochlore, including quenched phases (micron-scale pyrochlore crystals formed during quenching).
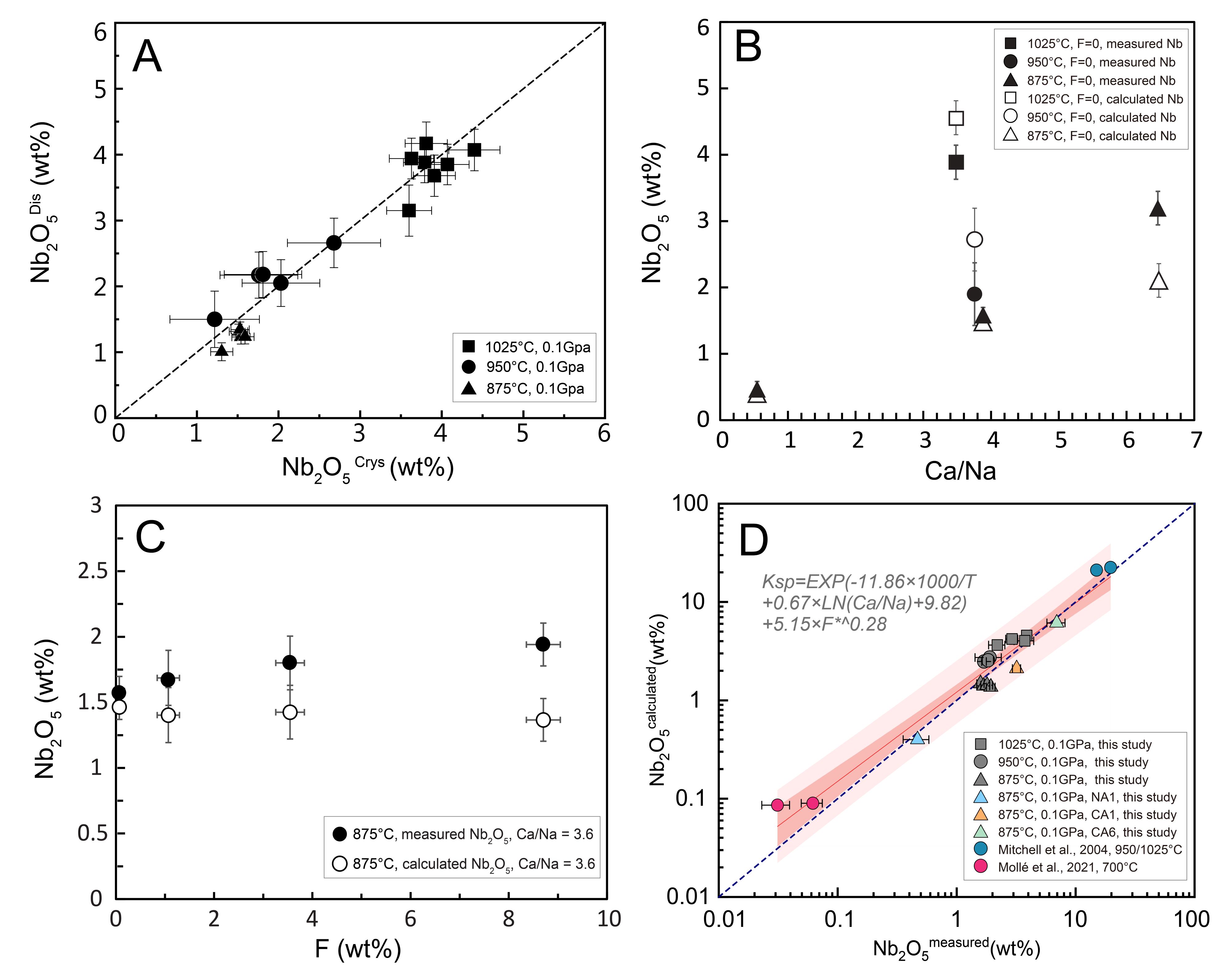
Figure 2. Melt compositions at saturation with pyrochlore and/or calciopyrochlore (A–C), and Nb concentrations calculated from a fractional crystallization model (D).
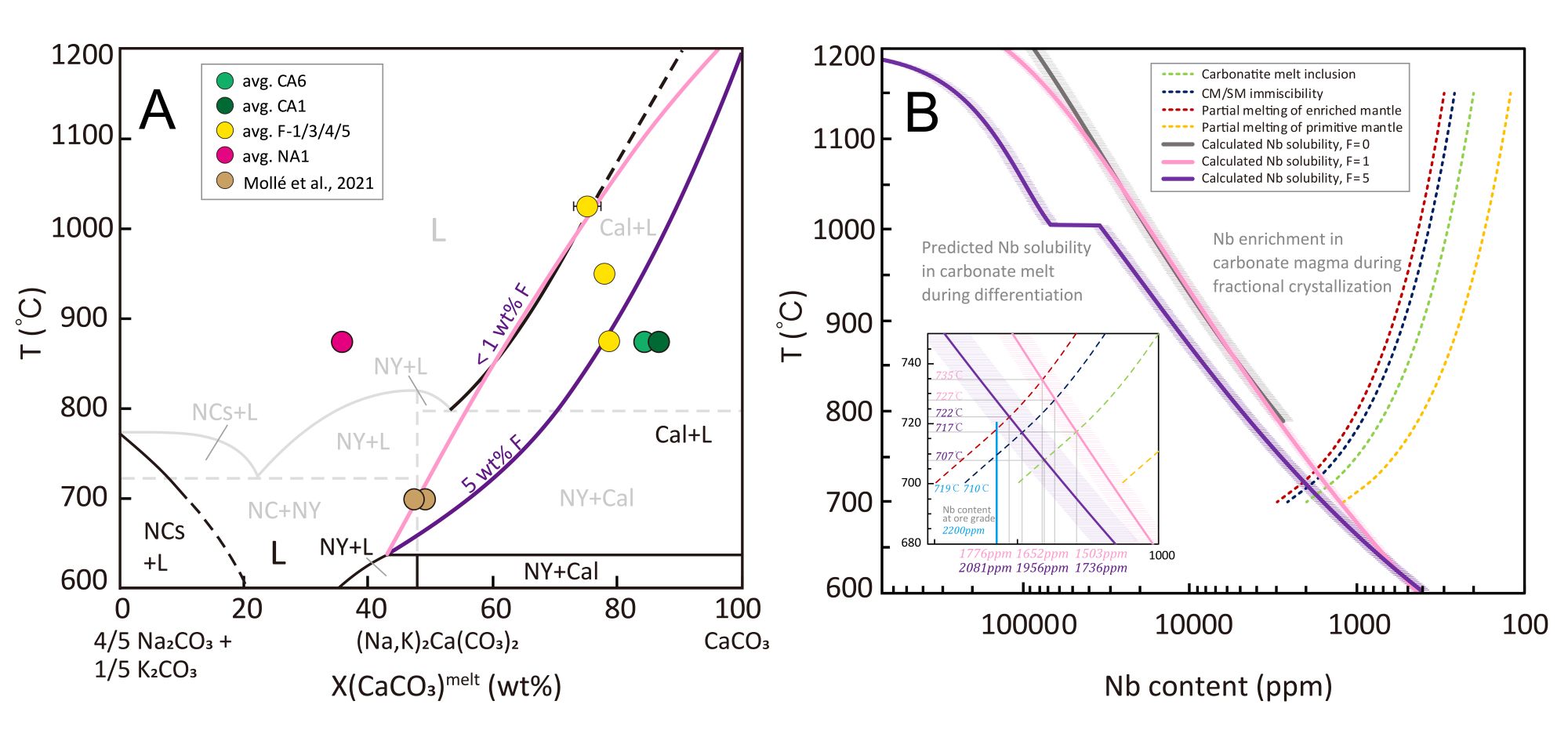
Figure 3. Carbonate phase diagram (A) and the sharp decrease in Nb solubility during carbonatite differentiation (B).

Figure 4. Schematic model for the formation of carbonatites and the mechanism of pyrochlore crystallization and Nb mineralization in carbonatite magma systems.

