Fluid inclusions are widely regarded as “time capsules” of geological fluids. By trapping tiny volumes of fluid during mineral growth, they can preserve information on temperature, pressure, chemical composition, and redox state, and thus provide a direct window into the formation and evolution of fluids in Earth’s interior. Molecular hydrogen (H2), one of the most reactive and mobile molecules in nature, has played a fundamental role in the evolution of the Earth system and life. Against the backdrop of the ongoing energy transition and a global “hydrogen rush,” understanding the genesis, migration, and accumulation of subsurface H2 has become a rapidly expanding frontier. In principle, H2-bearing fluid inclusions should offer critical constraints on these questions. In practice, however, the very properties that make H2 scientifically important (e.g., its small molecular size and high reactivity) also make it susceptible to diffusion and consumption, raising an urgent and fundamental concern: can H2-related signals in inclusions truly represent a geological legacy, or might they be inadvertently modified during laboratory analysis?
Raman spectroscopy is currently the most effective technique for identifying H2 in individual inclusions. Professor Xiaolin Wang’s team conducted systematic in situ Raman analyses of natural inclusions hosted by quartz, dolomite, and fluorite, together with synthetic inclusions prepared as fused silica capillary capsules (FSCCs) (Fig. 1). A key observation emerged across these systems: under laser irradiation, carbonaceous materials (CMs) (e.g., graphite, pyrobitumen, and diamond) can catalyze methane (CH4) decomposition and generate Raman-detectable H2 within seconds to tens of seconds (Fig. 2). When CO2 is present, carbon monoxide (CO) is also produced. Isotopic substitution experiments provide decisive evidence for the source of hydrogen: CD4 yields D2 rather than H2, confirming that hydrogen is derived from methane decomposition (Fig. 3). In 13CH4-bearing systems, 13C-graphite can be detected, indicating that graphite is the terminal carbon product of CH4 decomposition. Moreover, the newly formed 13C can react with 12CO2 to produce both 13CO and 12CO (Fig. 3), revealing coupled carbon-oxygen exchange pathways during irradiation.
The team further found that a pronounced H2 signal is only produced once the laser power exceeds a threshold (e.g., ~13.9 mW for a 532 nm laser) within typical analytical timescales, whereas the laser wavelength shows no strong control on H2 production across 473 nm, 532 nm, and 633 nm (Fig. 3). Based on these observations, the authors propose a mechanistic model for laser-induced CH4 decomposition on carbonaceous surfaces (Fig. 4). In CH4-only systems, carbon produced during decomposition progressively blankets reactive sites, suppressing sustained H2 generation. In contrast, in CO2-bearing systems, CO2 can “reactivate” these sites by consuming the newly formed carbon to produce CO, thereby preventing site passivation and allowing continued H2 production (Fig. 4). This model provides a unifying explanation for the stronger H2 signals observed in CO2-bearing inclusions.
These findings carry an important methodological implication: when Raman spectroscopy is used to characterize inclusion compositions, particularly in samples containing carbonaceous phases, laser power must be carefully controlled, and direct focusing on carbonaceous components should be avoided whenever possible, to minimize the risk of artificial H2 production. By reducing analytical artifacts, inclusion-based measurements can more reliably constrain deep-Earth fluid properties (T-P-x-fO2) and their evolution.
Beyond analytical guidance, the demonstrated reaction pathway is notable for its efficiency in very small reaction volumes and under modest thermal requirements. Together with the low cost and wide availability of carbonaceous materials (e.g., graphite and bitumen), these observations may provide useful perspectives for exploring methane-to-hydrogen conversion from a geoscience-informed viewpoint, while also highlighting the necessity of distinguishing laboratory-induced signals from genuine geological records.
This work was published in Geochimica et Cosmochimica Acta under the title “Hydrogen in natural inclusions: geological legacy or laboratory artifact?”. Ph.D. student Haokun Lian (School of Earth Sciences and Engineering, Nanjing University) is the first author, and Professor Xiaolin Wang is the corresponding author. Co-authors include Professor Matthew Steele-MacInnis (Department of Earth and Atmospheric Sciences, University of Alberta, Canada), Professor Wenxuan Hu and Ph.D. student Dongquan Sun (Nanjing University), and Professor I-Ming Chou (Institute of Deep-sea Science and Engineering, Chinese Academy of Sciences). This research was supported by the National Key Research and Development Program of China (2022YFF0800403), the National Natural Science Foundation of China (Grant Nos. 42173038 and 42130109), the Research Funds for the Frontiers Science Center for Critical Earth Material Cycling at Nanjing University, and the PetroChina Foundation Projects for Basic Scientific Research and Strategic Technology Reserves (2023D-5008-02).
Citation: Lian H., Wang X., Steele-MacInnis M., Sun D., Hu W., Chou I-M., Hydrogen in natural inclusions: geological legacy or laboratory artifact? Geochimica et Cosmochimica Acta, 2026. DOI: https://doi.org/10.1016/j.gca.2026.01.002

Fig. 1. Natural inclusions containing carbonaceous materials and methane in fluorite (Fl), dolomite (Dol), and quartz (Qtz), and fused silica capillary capsules (FSCC) loaded with gas and carbonaceous materials. Pyrbit = pyrobitumen, Gr = graphite.
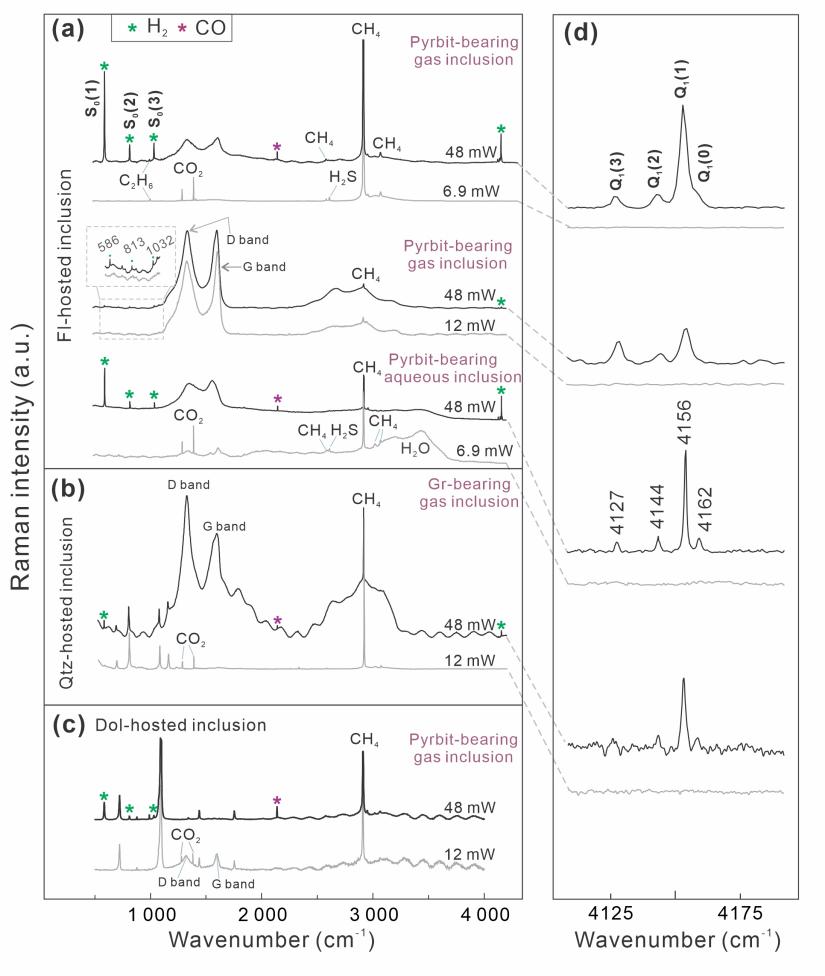
Fig. 2. H2 production in natural inclusions.
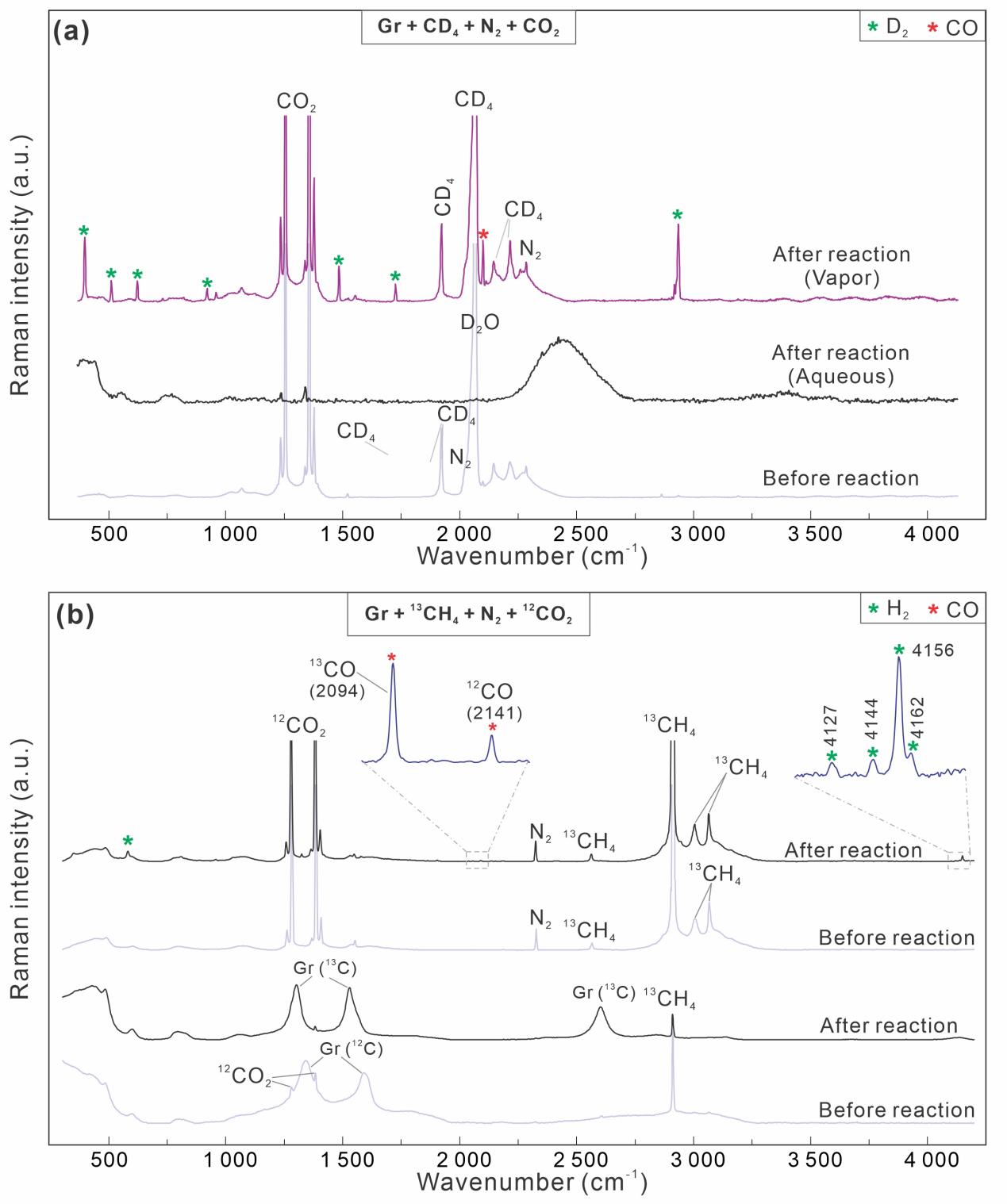
Fig. 3. H2 production in fused silica capillary capsules containing CD4/13CH4-Gr-CO2-N2 systems.
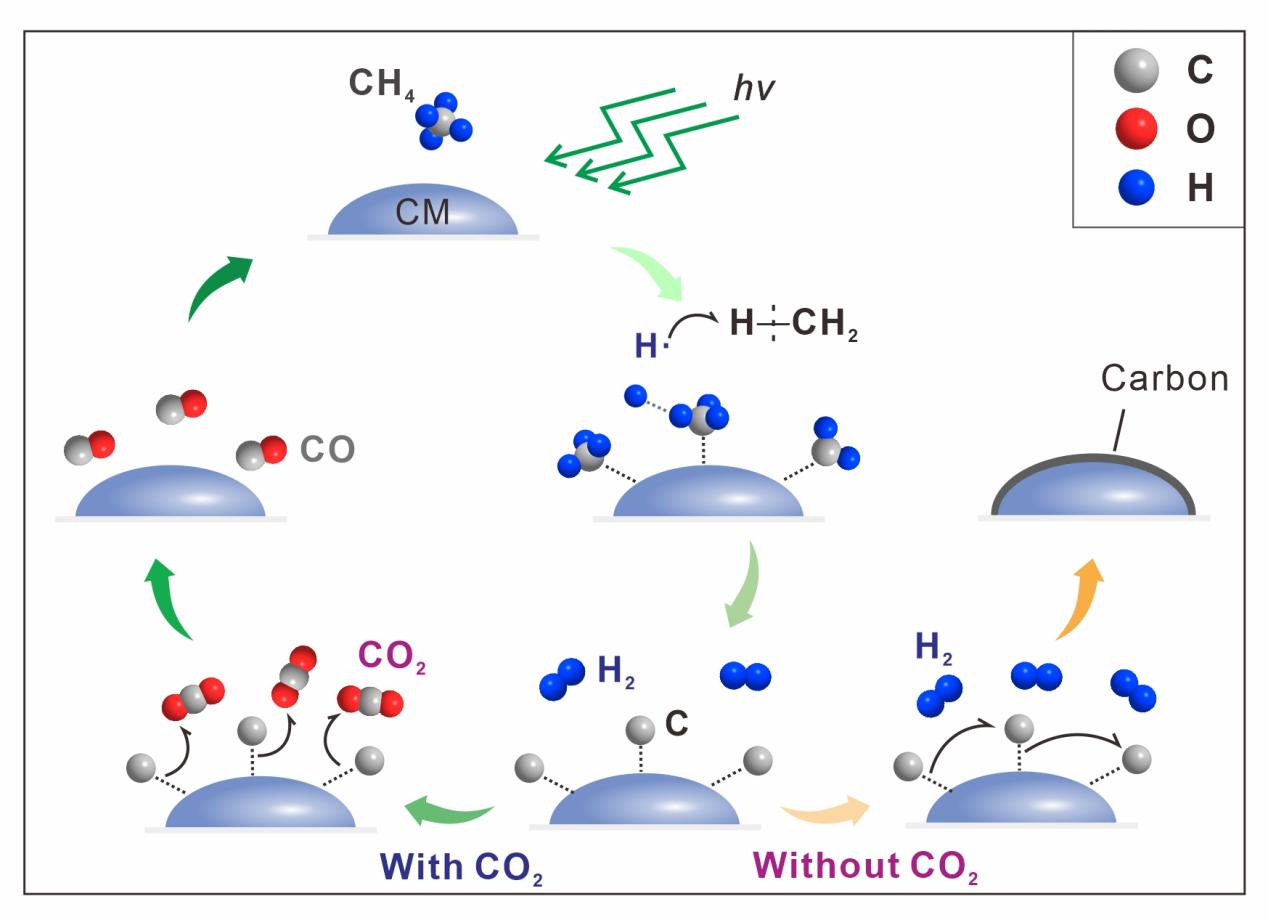
Fig. 4. Pathway of H2 production from CH4 under laser induction and catalysis by carbonaceous materials (CM).

